Radiation therapy for colorectal cancer
Doctors use different types of radiation therapy to treat both rectal and colon cancer. Being treated with chemotherapy at the same time can make radiation therapy more effective for some colorectal cancers.
What is radiation therapy? How does it work to treat cancer?
Radiation therapy is a form of cancer treatment that uses high-energy rays to destroy cancer cells. The cells in your body normally grow and divide to form new cells. Cancer cells grow and divide faster than normal cells, which can create tumors. Radiation damages the DNA inside cells. When the cells are damaged, they stop growing and dividing and eventually die.

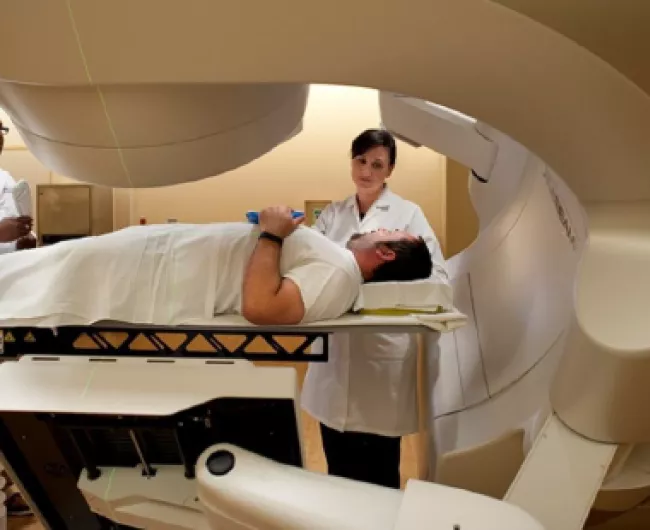
Radiation for rectal cancer
Radiation therapy is a common treatment for rectal cancer. It is used either before or after surgery, and usually along with chemotherapy.
It may be used prior to surgery (neoadjuvant therapy) to shrink a tumor to make it easier to remove, or afterwards to destroy cancer cells that cannot be removed with surgery.
Rectal tumors that come back in the pelvis after radiation therapy may be re-treated with radiation.
Radiation therapy may also be used to ease symptoms of advanced rectal cancer such as pain, bleeding, or intestinal obstructions.
Types of radiation
Side effects from radiation therapy
Talk with your doctor about the possible side effects of your radiation therapy regimen before your treatment begins so you know what to expect. Most side effects go away soon after treatment is finished, but some may not go away completely. Ask your doctor about any lasting side effects.
Possible side effects from radiation therapy for colorectal cancer may include:
- fatigue
- skin irritation, ranging from redness to blisters
- nausea
- diarrhea
- painful bowel movements
- bloody stools
- stool leakage (bowel incontinence)
- burning or pain while urinating
- urinary frequency
- blood in the urine
- sexual health issues (erectile dysfunction in men and vaginal irritation in women)
- infertility
Your fertility:
If you are planning to have a child, talk to your doctor before treatment begins about your options for preserving fertility.
More about treatment
ImmunotherapyTop resources

Christy Williams: Biomarker testing leads to successful treatment
Statistics suggested that Christy’s odds of survival were grim, so she leaned into her faith and kept a positive outlook. She tried to control what she could. And, critically, she received biomarker testing.

EPIC Act to Advance New CRC Therapies Needs Support
The EPIC Act would encourage investment in clinical trials for additional uses of existing drugs.

Michelle Cappel: Biomarker testing extends life
Michelle Cappel owes a lot to colorectal cancer biomarker testing — seven years of life and counting.





